Technology Paves Way for Non-Invasive Breast Cancer Treatment
Media Contact:
Julie Kiefer
Director, Research Communications, University of Utah Health
Email: Julie.Kiefer@hsc.utah.edu
Adapted from an article published by U of U Health Radiology and Imaging Sciences
A clinical trial at Huntsman Cancer Institute at the University of Utah is testing a technology developed at University of Utah Health that can kill breast tumors without surgery. The milestone marks a turning point in the evolution of less invasive methods for treating breast cancer.
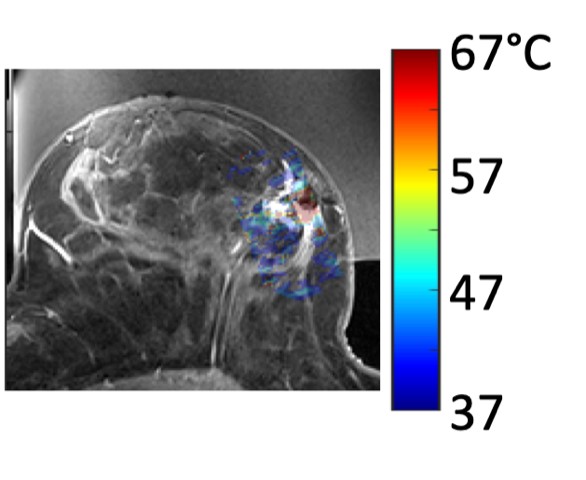
The MUSE Magnetic Resonance Guided Focused Ultrasound system uses magnetic resonance imaging (MRI) to locate and target the breast tumor, then high-intensity focused ultrasound heats the tissue and kills the cancer.

Allison Payne, Ph.D., associate professor in the Department of Radiology and Imaging Sciences and the Utah Center for Advanced Imaging Research (UCAIR) developed the technology with radiology faculty Dennis Parker, Ph.D., Rock Hadley, PhD, Henrik Odéen, PhD, and engineers Robb Merrill and Emilee Minalga. They made the first prototype in 2012 and refined the system over the next decade. Last year they received the approvals needed to evaluate the technology with breast cancer patients in a clinical trial.
“Our focused ultrasound lab is truly a global leader and innovator in developing and improving this clinical tool,” says Satoshi Minoshima, M.D., Ph.D., the Anne G. Osborn Chair of Radiology and Imaging Sciences.
Payne’s MUSE system is based around a special table she developed to help a woman feel comfortable for long periods of time in the MRI machine. The device delivers ultrasound waves into the tumor, focusing the energy into a point the size of a grain of rice. The energy heats up the cancerous tissue in 30 second intervals, moving to precisely target the entire three-dimensional space of the tumor. The patient is awake during the outpatient procedure, which could take up to a few hours.

The new, non-invasive approach to targeting breast tumors is currently being tested in a phase 1 clinical trial led by Cindy Matsen, M.D., a breast surgeon at Huntsman Cancer Institute (HCI) and an associate professor in the Department of Surgery. In the first phase feasibility study, which aims to enroll 34 participants, the MUSE system will ablate up to 50% of the tumor, then HCI surgeons remove the whole tumor. Pathologists will determine whether the system killed the tissue as planned.
“The clinical trial in no way impedes the patient’s standard of care. Patients that participate are generously giving their time so that we can evaluate this technology for future use,” explains Payne.
Eventually, researchers hope this treatment will progress through phase 3 clinical trials and replace surgery for some breast cancer patients. Payne predicts that it will take at least five years for MUSE to become available for commercial use.
Women interested in the trial can ask their doctor about participating in the study. Find more information at ClinicalTrials.gov.